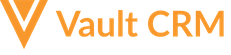Enabling Controlled Substance Sampling
Users can capture and validate controlled substance disbursement information on the call report, creating an auditable record and supporting compliance with regulations. In order to disburse samples or create sample requests/BRCs for controlled substances, users must have access to the appropriate product, address, and state credential records. When users attempt to save or submit a call report, DEA license information is validated to ensure the HCP is eligible to receive the selected controlled substance samples and BRCs. DEA license information is stored on HCP address records, but it is copied to the call report.
Validation Disclaimer
On the Windows (Beta) platform, this feature is released without validation for sampling. The feature requires enablement and does not affect other Vault CRM functionality. It was extensively tested by our QA team during the development and release process and will go through validation at a later date.
Who can use this feature?
- End Users — Browser, iPad, iPhone, Windows (Beta)
- Using Controlled Substance Sampling
- Users do not require an additional license
Configuring Controlled Substance Sampling for
To configure this feature:
- Ensure the following configuration is complete:
- Navigate to Admin > Users & Groups > Permission Sets.
- Select the appropriate permission set.
- Select the Objects tab.
- Ensure business admins have at least the following permissions (if not already granted in Configuring Call Sampling):
Object Object Permission Object Types Fields Field Permissions address__v
CRED
companymaintained__v
repmaintained__v
- dea__v
- dea_address__v
- dea_expiration_date__v
- dea_schedule__v
- dea_status__v
Edit
call2__v CRED call_report__v - address__v
- dea__v
- dea_expiration_date__v
- ship_dea__v
- ship_dea_expiration_date__v
Edit samples_state_credential_settings__v
CRED
n/a
- allowed_schedules__v
- credential__v
- state_province__v
Edit
sample_transaction__v
CRED
- adjustment__v
- disbursement__v
- receipt__v
- return__v
- transfer__v
- dea__v
- dea_expiration_date__v
Edit
To configure this feature:
- Ensure the following configuration is complete:
- Navigate to Admin > Users & Groups > Permission Sets.
- Select the appropriate permission set.
- Select the Objects tab.
-
Ensure end users have the following permissions (if not already granted in Configuring Call Sampling):
Object Object Permission Object Types Fields Field Permissions address__v
R
repmaintained__v
- dea__v
- dea_address__v
- dea_expiration_date__v
- dea_schedule__v
- dea_status__v
Read
call2__v
CRE
call_report__v - address__v
- dea__v
- dea_expiration_date__v
Edit
samples_state_credential_settings__v
R
n/a
All fields Read
sample_transaction__v
R
- adjustment__v
- disbursement__v
- receipt__v
- return__v
- transfer__v
- dea__v
- dea_expiration_date__v
Read
- Navigate to Business Admin > Objects > veeva_settings__v.
- Select the enable_dual_license_check__v Veeva Setting checkbox (optional). This ensures validation runs on both the HCP’s DEA license and state license, if configured.
- Navigate to Business Admin > Objects > vmobile_object_configuration__v.
- Ensure the appropriate VMOCs are enabled for the samples_state_credential_settings__v object.
Record Updates for Controlled Substance Sampling
As part of enabling controlled substance sampling, update address__v and product__v records, and create samples_state_credential_settings__v records.
- Navigate to Business Admin > Objects > address__v.
- Populate the following fields on the appropriate accounts’ address__v records:
- dea__v - Populate with the HCP's DEA license number
- dea_address__v - Select the checkbox to indicate this is a DEA address
- dea_expiration_date__v - Populate with the HCP's DEA license expiration date
- dea_schedule__v - Populate with a comma-delimited list of the schedules the account can receive for the designated DEA License
Ensure the text for DEA schedules is consistent across all areas where schedules are specified. In order for an account to receive a product, the schedule associated to the controlled substance sample must be an exact, literal match to one of the schedules listed in the dea_schedule__v field.
- dea_status__v - Set the license status to either Valid or Invalid. Valid indicates the HCP can receive controlled substance samples, while invalid indicates they cannot receive controlled substance samples.
Permission for dea_license_address__v field on the address__v object is not required. The DEA Address fields on the call report are copied from name__v (Address Line 1), address_line_2__v, city__v, state_province__v, zip__v, and zip_4__v on the address__v record, not from the dea_license_address__v field.
If the address record is locked, only administrators or users with Modify All permissions can edit these fields.
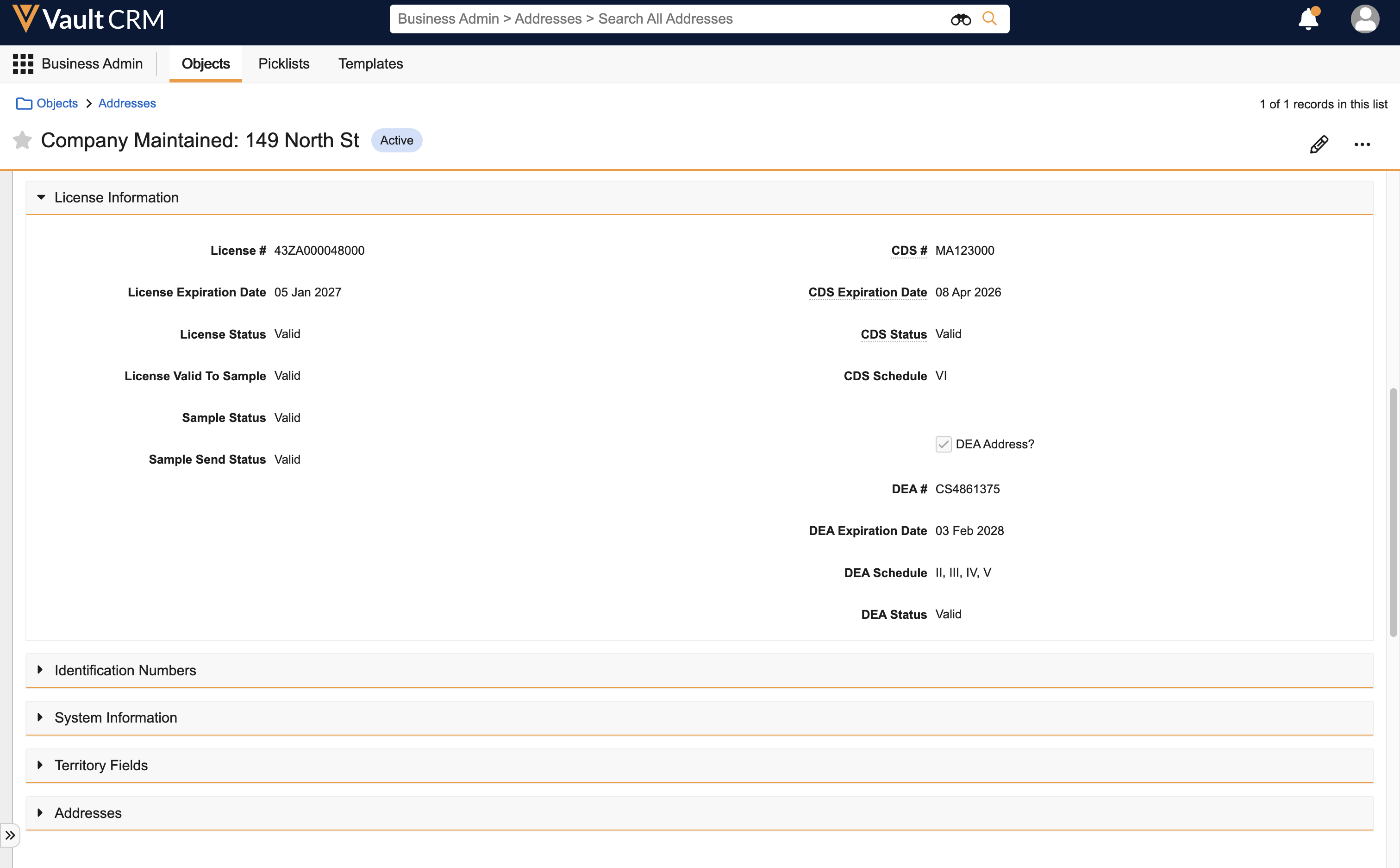
- Navigate to Business Admin > Objects > product__v.
- Populate the following fields on the appropriate product__v records:
- controlled_substance__v - Select this checkbox to mark the product as a controlled substance
- schedule__v - Populate this field with the appropriate schedule for the product. Text in the Schedule field must be consistent across all areas where DEA schedules are specified; sample validation requires exact textual matches for this field.
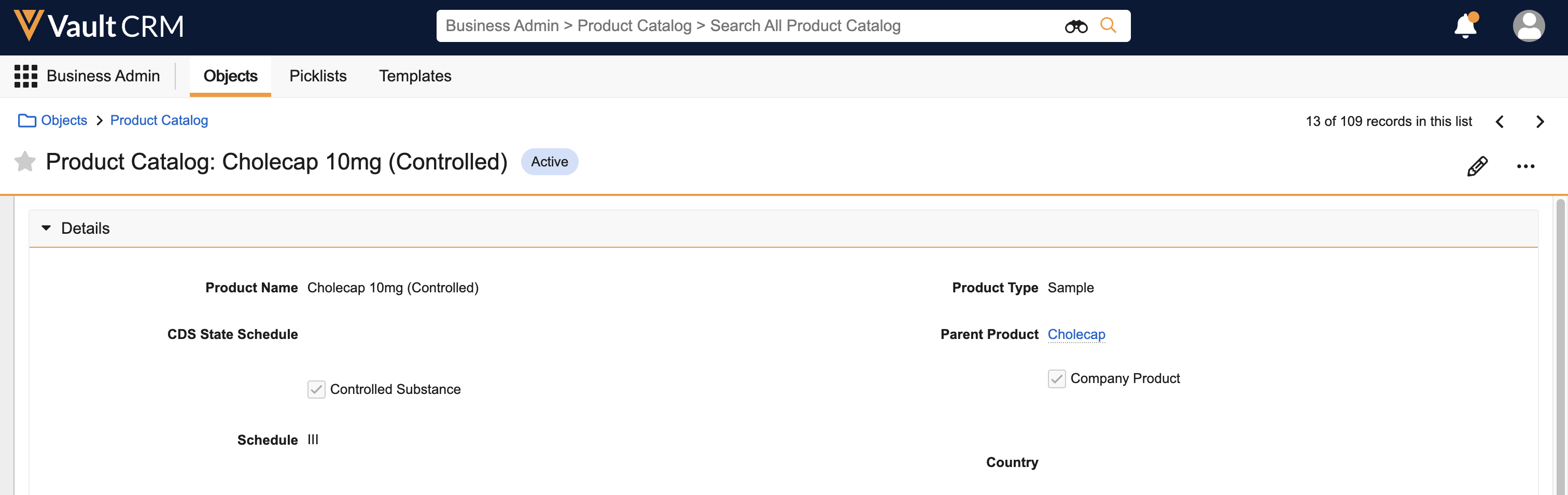
- Navigate to Business Admin > Objects > samples_state_credential_settings__v.
- Create the appropriate samples_state_credential_settings__v records. In order to receive controlled substance samples, there must be a samples_state_credential_settings__v record matching the HCP's credentials and state, and the product schedule of the sample being disbursed. Ensure all appropriate states have samples_state_credential_settings__v records for the credential levels where HCPs are authorized to receive controlled substance samples.
- Populate the following fields on the samples_state_credential_settings__v records:
- credential__v - The appropriate HCP credential level to allow controlled substance sampling
- allowed_schedules__v - Controlled substance schedules HCPs with the selected credentials are authorized to receive
If there are existing samples_state_credential_settings__v records where the restricted_products__v field is populated for Restricting Sampling by Account Credentials and State, create separate records for controlled substance sampling. The allowed_schedules__v and restricted_products__v fields cannot both be populated on the same record.
-
state_province__v - State where HCPs with the selected credentials are authorized to receive controlled substance samples. The values in the state_province__v field on the samples_state_credential_settings__v object must match picklist value API names from the state_province__v field on the address__v object.
Country restrictions are not available when the allowed_schedules__v field is populated. The state_province__v field must be used.
Optional Layout Configuration
Optionally, admins can enable a DEA status indicator, and/or display of DEA license information on the call report.
- Navigate to Admin > Users & Groups > Permission Sets.
- Select the appropriate permission set.
- Select the Objects tab.
- Grant users access to the address_dea_status__v field control on the call2__v object.
- Navigate to Admin > Configuration > Objects > call2__v > Layouts.
- Place the address_dea_status__v field control on the appropriate call2__v layouts (enables a DEA status indicator on the call report). Do not place the control in a section control section.
-
Place the following fields on the appropriate call2__v layouts (displays DEA license information on the call report):
- dea__v
- dea_expiration_date__v
Configuring Ship-To DEA License Information for
Users must also capture DEA license information for sample requests/BRCs of controlled substances. To enable sample requests/BRCs of controlled substances, complete the following additional configuration:
- Navigate to Admin > Users & Groups > Permission Sets.
- Select the appropriate permission set.
- Select the Objects tab.
- Ensure end users creating sample requests/BRCs have the following additional field permissions:
Object Object Permission Object Types Fields Field Permissions call2__v
CRED call_report__v - ship_dea__v
- ship_dea_expiration_date__v
Edit
- Navigate to Admin > Configuration > Objects > call2__v > Layouts.
-
Add the following fields to the appropriate layouts on the call2__v object (optional; displays DEA license information on the call report):
- ship_dea__v
- ship_dea_expiration_date__v
Using Controlled Substance Sampling as
If an HCP is licensed to receive controlled substances, users add controlled substance sample products to the call report through the same controls as regular sample products/BRCs. See Selecting Products for more information.
Validating DEA License Information
Additional validation helps ensure disbursements of controlled substances comply with the more stringent regulations for controlled substance sampling. In addition to the default validation criteria for Saving a Call with Samples, Validating Samples on Signature Capture, and Submitting a Call with Samples, controlled substance sampling must meet the following requirements:
- The call address/ship-to address and the DEA Address for the selected person account must match exactly
- DEA information for the selected person account address must meet the following criteria:
- DEA License Number - The DEA license number for the selected address must follow a specific format, which is enforced via an algorithm check when the account signs for controlled substances
- DEA Expiration Date - The expiration date must be greater than or equal to the current date
- DEA Schedule - The schedule associated to the controlled substance sample must be an exact, literal match to one of the schedules listed in the dea_schedule__v field on the account’s address__v record
- DEA Status - The DEA Status must be set to Valid
- There must be a samples_state_credential_settings__v record meeting the following criteria:
- Allowed Schedules - The product schedule of any samples/BRCs on the call report must be included in the allowed_schedules__v list
- Credentials - The selected credentials must match the signing account's credentials
- State - The state/province must match the call address or ship-to address state/province
- If the enable_dual_license_check__v Veeva Setting is selected, the HCP must have valid state license information for the call addresses’ state
- If Samples State Settings are enabled, the state and delivery mechanism for the sample/BRC cannot be included in the list of restrictions
Swapping the Signee
When controlled substances are on the call report, the Swapping the Signee feature only allows users to select a new signee whose DEA Address information is an exact, literal match to the previous signee’s DEA Address information.
Using the DEA Sample Status Indicator
If the address_dea_status__v field control is added to the call report layout, DEA status indicators display for accounts and in the appropriate Address picklists on the call report. This gives users a visual reminder of whether or not the HCP can receive controlled substance samples for the specific address. DEA status indicators only display for person accounts.